Velux Basic Blackout Blind

Velux DBL Budget Blackout Blinds suitable for VELUX roof windows manufactured before 2014, the VELUX budget blind is the perfect budget solution for replacing your old tired blinds. The blinds are slightly more bulkier than the slimline standard blackout blind. And they also emit light around the frame so not total blackout. 2 - year guarantee.
Claim VELUX Rewards on EVERY purchase of a VELUX blind, you can claim from £3 - £40 rewards for EACH product EVERY time. Homeowners and installers are eligible to collect VELUX Rewards

Mileage Calculator
For installations, there is a one off charge based on your location. This is a single charge, not related to the number of items being installed.
Use the form below to enter your details and we will calculate the mileage cost.
 calculating, please wait...
calculating, please wait...
1Choose a colour/pattern
Standard Colours
more options ▼ 2Window Type need help?
- ggl Centre-pivot GGL
- ggu White Polyurethane Centre-pivot
- gpl Top-hung GPL
- gpu White Polyurethane Top-hung
- ghl Pine Finish Top-hung GHL
- ghu White Polyurethane Top-hung GHU
more options ▼ 3Window Size need help?
-
c02
C02 (mfd Apr 01 - Feb 14)
£35.14
£57.60 -
c04
C04 (mfd Apr 01 - Feb 14)
£38.06
£62.40 -
f04
F04 (mfd Apr 01 - Feb 14)
£42.46
£69.60 -
f06
F06 (mfd Apr 01 - Feb 14)
£45.38
£74.40 -
m04
M04 (mfd Apr 01 - Feb 14)
£43.92
£72.00 -
m06
M06 (mfd Apr 01 - Feb 14)
£46.85
£76.80 -
m08
M08 (mfd Apr 01 - Feb 14)
£49.78
£81.60 -
m10
M10 (mfd Apr 01 - Feb 14)
£55.63
£91.20 -
p06
P06 (mfd Apr 01 - Feb 14)
£51.24
£84.00 -
p08
P08 (mfd Apr 01 - Feb 14)
£55.63
£91.20 -
s06
S06 (mfd Apr 01 - Feb 14)
£58.56
£96.00 -
s08
S08 (mfd Apr 01 - Feb 14)
£61.49
£100.80 -
u04
U04 (mfd Apr 01 - Feb 14)
£60.02
£98.40 -
u08
U08 (mfd Apr 01 - Feb 14)
£68.81
£112.80 -
1
1 (mfd before May 92 )
£43.92
£72.00 -
2
2 (mfd before May 92 )
£49.78
£81.60 -
4
4 (mfd before May 92 )
£58.56
£96.00 -
6
6 (mfd before May 92 )
£38.06
£62.40 -
7
7 (mfd before May 92 )
£60.02
£98.40 -
8
8 (mfd before May 92 )
£68.81
£112.80 -
304
304 (mfd May 92 - Apr 01)
£43.92
£72.00 -
306
306 (mfd May 92 - Apr 01)
£46.85
£76.80 -
308
308 (mfd May 92 - Apr 01)
£49.78
£81.60 -
310
310 (mfd May 92 - Apr 01)
£55.63
£91.20 -
406
406 (mfd May 92 - Apr 01)
£51.24
£84.00 -
408
408 (mfd May 92 - Apr 01)
£55.63
£91.20 -
606
606 (mfd May 92 - Apr 01)
£58.56
£96.00 -
608
608 (mfd May 92 - Apr 01)
£61.49
£100.80 -
804
804 (mfd May 92 - Apr 01)
£60.02
£98.40 -
808
808 (mfd May 92 - Apr 01)
£68.81
£112.80
-
c02
C02 (mfd Apr 01 - Feb 14)
£35.14
£57.60 -
c04
C04 (mfd Apr 01 - Feb 14)
£38.06
£62.40 -
f06
F06 (mfd Apr 01 - Feb 14)
£45.38
£74.40 -
m04
M04 (mfd Apr 01 - Feb 14)
£43.92
£72.00 -
m06
M06 (mfd Apr 01 - Feb 14)
£46.85
£76.80 -
m08
M08 (mfd Apr 01 - Feb 14)
£49.78
£81.60 -
m10
M10 (mfd Apr 01 - Feb 14)
£55.63
£91.20 -
p06
P06 (mfd Apr 01 - Feb 14)
£51.24
£84.00 -
p08
P08 (mfd Apr 01 - Feb 14)
£55.63
£91.20 -
s06
S06 (mfd Apr 01 - Feb 14)
£58.56
£96.00 -
u04
U04 (mfd Apr 01 - Feb 14)
£60.02
£98.40 -
u08
U08 (mfd Apr 01 - Feb 14)
£68.81
£112.80
-
c04
C04 (mfd Apr 01 - Feb 14)
£38.06
£62.40 -
f06
F06 (mfd Apr 01 - Feb 14)
£45.38
£74.40 -
m04
M04 (mfd Apr 01 - Feb 14)
£43.92
£72.00 -
m06
M06 (mfd Apr 01 - Feb 14)
£46.85
£76.80 -
m08
M08 (mfd Apr 01 - Feb 14)
£49.78
£81.60 -
m10
M10 (mfd Apr 01 - Feb 14)
£55.63
£91.20 -
p06
P06 (mfd Apr 01 - Feb 14)
£51.24
£84.00 -
p08
P08 (mfd Apr 01 - Feb 14)
£55.63
£91.20 -
s06
S06 (mfd Apr 01 - Feb 14)
£58.56
£96.00 -
s08
S08 (mfd Apr 01 - Feb 14)
£61.49
£100.80 -
u04
U04 (mfd Apr 01 - Feb 14)
£60.02
£98.40 -
u08
U08 (mfd Apr 01 - Feb 14)
£68.81
£112.80 -
304
304 (mfd May 92 - Apr 01)
£43.92
£72.00 -
306
306 (mfd May 92 - Apr 01)
£46.85
£76.80 -
308
308 (mfd May 92 - Apr 01)
£49.78
£81.60 -
310
310 (mfd May 92 - Apr 01)
£55.63
£91.20 -
406
406 (mfd May 92 - Apr 01)
£51.24
£84.00 -
408
408 (mfd May 92 - Apr 01)
£55.63
£91.20 -
606
606 (mfd May 92 - Apr 01)
£58.56
£96.00 -
608
608 (mfd May 92 - Apr 01)
£61.49
£100.80 -
804
804 (mfd May 92 - Apr 01)
£60.02
£98.40 -
808
808 (mfd May 92 - Apr 01)
£68.81
£112.80
-
c04
C04 (mfd Apr 01 - Feb 14)
£38.06
£62.40 -
f06
F06 (mfd Apr 01 - Feb 14)
£45.38
£74.40 -
m04
M04 (mfd Apr 01 - Feb 14)
£43.92
£72.00 -
m06
M06 (mfd Apr 01 - Feb 14)
£46.85
£76.80 -
m08
M08 (mfd Apr 01 - Feb 14)
£49.78
£81.60 -
m10
M10 (mfd Apr 01 - Feb 14)
£55.63
£91.20 -
p06
P06 (mfd Apr 01 - Feb 14)
£51.24
£84.00 -
p08
P08 (mfd Apr 01 - Feb 14)
£55.63
£91.20 -
s06
S06 (mfd Apr 01 - Feb 14)
£58.56
£96.00 -
u04
U04 (mfd Apr 01 - Feb 14)
£60.02
£98.40 -
u08
U08 (mfd Apr 01 - Feb 14)
£68.81
£112.80
-
c04
C04 (mfd Apr 01 - Feb 14)
£38.06
£62.40 -
f06
F06 (mfd Apr 01 - Feb 14)
£45.38
£74.40 -
m04
M04 (mfd Apr 01 - Feb 14)
£43.92
£72.00 -
m06
M06 (mfd Apr 01 - Feb 14)
£46.85
£76.80 -
m08
M08 (mfd Apr 01 - Feb 14)
£49.78
£81.60 -
m10
M10 (mfd Apr 01 - Feb 14)
£55.63
£91.20 -
p06
P06 (mfd Apr 01 - Feb 14)
£51.24
£84.00 -
p08
P08 (mfd Apr 01 - Feb 14)
£55.63
£91.20 -
s06
S06 (mfd Apr 01 - Feb 14)
£58.56
£96.00 -
u04
U04 (mfd Apr 01 - Feb 14)
£60.02
£98.40 -
u08
U08 (mfd Apr 01 - Feb 14)
£68.81
£112.80 -
1
1 (mfd before May 92 )
£43.92
£72.00 -
2
2 (mfd before May 92 )
£49.78
£81.60 -
4
4 (mfd before May 92 )
£58.56
£96.00 -
6
6 (mfd before May 92 )
£38.06
£62.40 -
7
7 (mfd before May 92 )
£60.02
£98.40 -
8
8 (mfd before May 92 )
£68.81
£112.80 -
304
304 (mfd May 92 - Apr 01)
£43.92
£72.00 -
306
306 (mfd May 92 - Apr 01)
£46.85
£76.80 -
308
308 (mfd May 92 - Apr 01)
£49.78
£81.60 -
310
310 (mfd May 92 - Apr 01)
£55.63
£91.20 -
406
406 (mfd May 92 - Apr 01)
£51.24
£84.00 -
408
408 (mfd May 92 - Apr 01)
£55.63
£91.20 -
606
606 (mfd May 92 - Apr 01)
£58.56
£96.00 -
608
608 (mfd May 92 - Apr 01)
£61.49
£100.80 -
804
804 (mfd May 92 - Apr 01)
£60.02
£98.40 -
808
808 (mfd May 92 - Apr 01)
£68.81
£112.80
-
c04
C04 (mfd Apr 01 - Feb 14)
£38.06
£62.40 -
f06
F06 (mfd Apr 01 - Feb 14)
£45.38
£74.40 -
m04
M04 (mfd Apr 01 - Feb 14)
£43.92
£72.00 -
m06
M06 (mfd Apr 01 - Feb 14)
£46.85
£76.80 -
m08
M08 (mfd Apr 01 - Feb 14)
£49.78
£81.60 -
p06
P06 (mfd Apr 01 - Feb 14)
£51.24
£84.00 -
p08
P08 (mfd Apr 01 - Feb 14)
£55.63
£91.20 -
s06
S06 (mfd Apr 01 - Feb 14)
£58.56
£96.00 -
s08
S08 (mfd Apr 01 - Feb 14)
£61.49
£100.80
Help & Useful Information
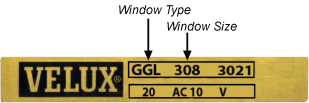
How do I find which Window Type I have?
VELUX Type Signs (Data plate) explained
Every VELUX roof window carries a unique Type Sign that details the type and size of the window. In most cases the Type Sign is located at the top right-hand of the sash, behind the control bar.
Type Signs differ in appearance depending on the model of your VELUX window. Please identify and select the version of Type Sign on your window from the illustrations below:
Step 1 - Locating the type sign.
In most cases the Type Sign is located at the top right-hand of the sash, behind the control bar.

Step 2 - Finding the codes.
Depending on the year your window was produced you'll find one of the type signs listed below.